The Difference Between Metals and Nonmetals.
Metals and nonmetals share similarities on a fundamental level. Electrons, protons and neutrons compose all members of both groups. Similarly, all elements can react, change state and form compounds, although some do so more readily than others.
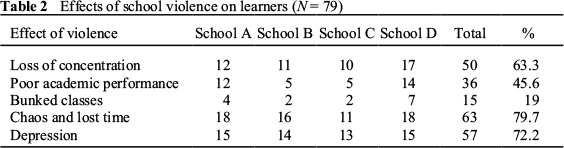
Notice that metals and non-metals have opposite properties to each other. It is usually easy to tell metals and non-metals apart, but some tests are more reliable than others. For example, using a.
On Metals Nonmetals And Metalloid. Chemical Prperties of Metals Most metals are chemically reactive, reacting with oxygen in the air to form oxides over changing timescales (for example iron rusts over years and potassium burns in seconds). The alkali metals react quickest followed by the alkaline earth metals, found in the leftmost two groups of the periodic table.
Compare and contrast the properties of metals, metalloids, and nonmetals. -Metals- good conductors of heat and electricity and reflect light and heat, most luster (shine) and most are malleable (hammered or rolled into sheets) -Non-metals- poor conductors of heat and electricity, most are gas at room temperature, those that are solid are not.
There are a number of ways in which a metal can be differientiate a metal from a non-metals, these involve contrasting the properties between metals and non-metals and testing them to distinguish.
Most elements are metals, but quite a few are nonmetals.It's important to be able to distinguish between the different types of elements. Here are lists of five metals and five nonmetals, an explanation of how you can tell them apart, and some examples of their uses.
The main difference between metals and non-metals is Metals are electropositive in nature as they easily lose electrons, so they are reducing agents. On the contrary, Non-metals are electronegative because they gain electrons and thus they are oxidising agents.